Buffer capacity | Acids and bases | AP Chemistry | Khan Academy

Author: Khan Academy via YouTube
Go to Source

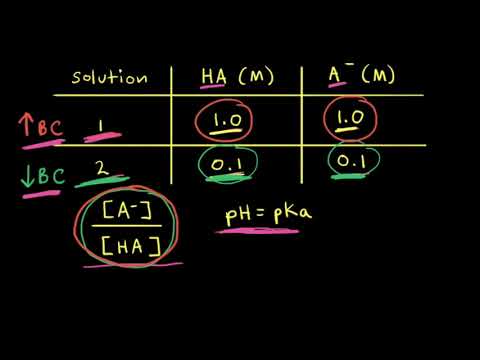
The capacity of a buffer to neutralize added acid or base depends on the concentrations of HA and A⁻ in solution. For a given ratio of [HA] to [A⁻], the greater the concentrations, the higher the overall buffer capacity. When [HA] is greater than [A⁻], the capacity is higher for added base than acid. When [A⁻] is greater than [HA], the capacity is higher for added acid than base. View more lessons or practice this subject at https://www.khanacademy.org/science/ap-chemistry-beta/x2eef969c74e0d802:acids-and-bases/x2eef969c74e0d802:buffers/v/buffer-capacity-ap
Khan Academy is a nonprofit organization with the mission of providing a free, world-class education for anyone, anywhere. We offer quizzes, questions, instructional videos, and articles on a range of academic subjects, including math, biology, chemistry, physics, history, economics, finance, grammar, preschool learning, and more. We provide teachers with tools and data so they can help their students develop the skills, habits, and mindsets for success in school and beyond. Khan Academy has been translated into dozens of languages, and 15 million people around the globe learn on Khan Academy every month. As a 501(c)(3) nonprofit organization, we would love your help!
Donate or volunteer today! Donate here: https://www.khanacademy.org/donate?utm_source=youtube&utm_medium=desc
Volunteer here: https://www.khanacademy.org/contribute?utm_source=youtube&utm_medium=desc
